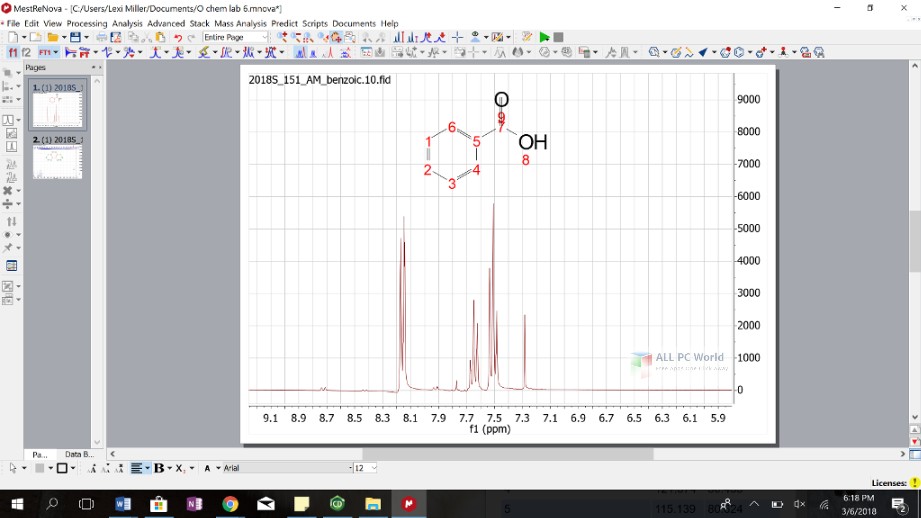 
(3) Additionally, TMS can be used as an external reference utilizing a sealed capillary tube of solvent+TMS and can be placed inside of the NMR tube and therefore not interact with your solute. The chemical shift of this singlet is also set to be δ 0 in the 13C spectrum, and all other chemical shifts are determined relative to it. In a fully decoupled 13C NMR spectrum, the carbon in the tetramethylsilane appears as a singlet, allowing for easy identification. 
Tetramethylsilane (TMS) became the established internal reference compound for NMR because it has a strong, sharp resonance line from its 12 protons, with a chemical shift at low resonance frequency relative to almost all other 1H resonances. Shimming will occur best with this sample size. Ī few useful sample tips: (1) All instruments are calibrated based on samples of 600 microliters.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
